
MHRA publishes regulatory roadmap for development of medical devices
The MHRA has published its regulatory roadmap detailing how new regulations for medical devices are to be rolled out across the UK.
Back to Top

The MHRA has published its regulatory roadmap detailing how new regulations for medical devices are to be rolled out across the UK.

Host Jordan Sollof is joined by Shuri Network members Tolu Awe and Felicia Akubue to highlight the benefits of being a part of the bursary programme.

Today’s briefing includes a new cloud-based platform to recruit retired doctors back to the NHS and an AI system to predict pancreatic cancer.
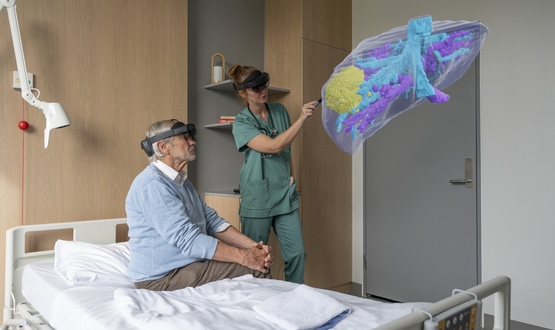
HoloCare’s surgical hologram technology is to be deployed at Leeds Teaching Hospitals NHS Trust to help surgeons perform more efficiently and accurately.

Digital Health Rewired 2024 will feature a live AI “bake-off”, where a panel of NHS doctors will test AI LLM models on a series of diagnostic questions.

Three digital health companies focused on providing support to women during menopause have advanced to the CivTech Round 9 Accelerator.

This briefing includes solutions reaching the Accelerator stage of CivTech Accelerator Round 9 and The Breathing App launching an app for diabetes patients.

Orion Health is close to returning to profitability and is pivoting to “a number of

Fitbit confirmed its products would continue to remain platform-agnostic across both Android and iOS devices,
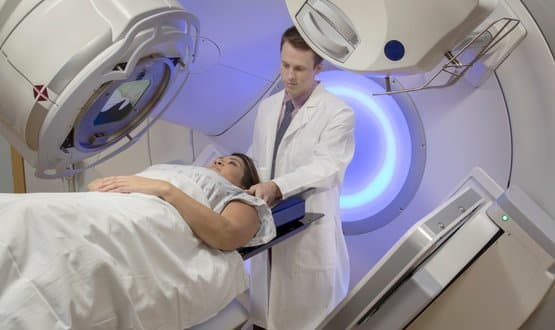
Cancer Research UK RadNet marks the charities largest ever investment in radiotherapy research and aims

The wireless cuff is designed to be used in any location alongside an app, OMRONConnect,

The funding will also be used to connect patient history and replace outdated paper systems,
