
Exclusive: Huma receives US FDA Class II clearance for SaMD platform
A Software-as-a-Medical-Device platform from Huma has received US Food and Drug Administration Class II clearance, boosting innovation in digital health.
Back to Top

A Software-as-a-Medical-Device platform from Huma has received US Food and Drug Administration Class II clearance, boosting innovation in digital health.

CEPI is providing $1.6m to establish a proof-of-concept for the aVaxziPen needle-free vaccine delivery platform to eliminate the need for frozen storage.

Voting opens today for elections to three professional advisory panels representing Digital Health Networks, a community of more than 7,000 NHS IT leaders.

The NHS plans to set up an academy to train new data and analytics specialists and could look to the private sector to fund the training.

A new report has been published looking at the benefits of more widespread adoption of cloud technology within the healthcare sector.
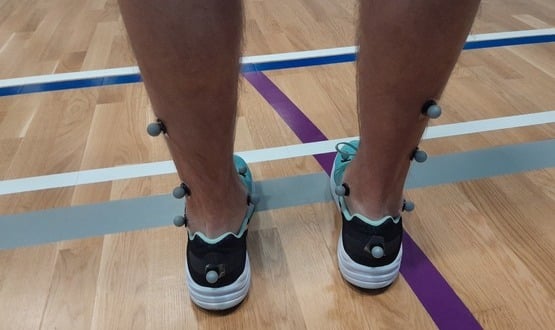
Performance capture technology used in Hollywood films is being researched for the benefits it could bring to rehabilitation for stroke patients.

Digital Health’s monthly roundup of contacts and go lives features Great Ormond Street and Roche UK partnering to improve children’s care using AI.

Milton Keynes University Hospital has implemented a digital FGM solution, which allows clinicians to view

Trails will enable staff to access the information through the summary care record application,with integration
Future funding for LHCREs may be redirected to fund other projects, including last week’s £1.8bn

The Asthma+me App allows patients to input information about their asthma and link up their

The Government has launched a review to explore how data and technology can deliver a
